(Acetyloxy)triphenylstannane (CHEM001081)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2009-06-19 21:58:31 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2016-11-09 01:08:26 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM001081 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | (Acetyloxy)triphenylstannane | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Nonsystemic fungicide (Acetyloxy)triphenylstannane belongs to the family of Aromatic Homomonocyclic Compounds. These are aromatic compounds containig only one ring, which is homocyclic. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
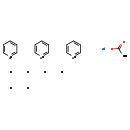
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C20H24O2Sn | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 415.120 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 416.080 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 900-95-8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | fentin acetate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | fentin acetate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | [H].[H].[H].[SnH3+].CC([O-])=O.[C]1=CC=CC=C1.[C]1=CC=CC=C1.[C]1=CC=CC=C1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/3C6H5.C2H4O2.Sn.6H/c3*1-2-4-6-5-3-1;1-2(3)4;;;;;;;/h3*1-5H;1H3,(H,3,4);;;;;;;/q;;;;+1;;;;;;/p-1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | JCKWWPISVOUYMM-UHFFFAOYSA-M | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as benzene and substituted derivatives. These are aromatic compounds containing one monocyclic ring system consisting of benzene. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Benzenoids | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Benzene and substituted derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Benzene and substituted derivatives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Oral (5) ; inhalation (5) ; dermal (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Organotin compounds produce neurotoxic and immunotoxic effects. Organotins may directly activate glial cells contributing to neuronal cell degeneration by local release of pro-inflammatory cytokines, tumor necrosis factor-_, and/or interleukins. They may also induce apoptosis by direct action on neuronal cells. Organotin compounds stimulate the neuronal release of and/or decrease of neuronal cell uptake of neurotransmitters in brain tissue, including aspartate, GABA, glutamate, norepinephrine, and serotonin. This may be either a contributing factor to or result of the neuronal cell loss. The immunotoxic effects of organotins are characterized by thymic atrophy caused by the suppression of proliferation of immature thymocytes and apoptosis of mature thymocytes. Organotin compounds are believed to exert these effects by suppressing DNA and protein synthesis, inducing the expression of genes involved in apoptosis (such as nur77), and disrupting the regulation of intracellular calcium levels, giving rise to the uncontrolled production of reactive oxygen species, release of cytochrome c to the cytosol, and the proteolytic and nucleolytic cascade of apoptosis. The suppression of proliferation of immature thymocytes further results in the suppression of T-cell-mediated immune responses. Organotins are also endocrine disruptors and are believed to contribute to obesity by inappropriate receptor activation, leading to adipocyte differentiation. Inorganic tin triggers eryptosis, contributing to tin-induced anemia. (5, 1, 2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Though tin metal is very poorly absorbed, tin compounds may be absorbed via oral, inhalation, or dermal routes, with organotin compounds being much more readily absorbed than inorganic tin compounds. Tin may enter the bloodstream and bind to hemoglobin, where it is distributed and accumulates mainly in the kidney, liver, lung, and bone. Organotin compounds may undergo dealkylation, hydroxylation, dearylation, and oxidation catalyzed by cytochrome P-450 enzymes in the liver. The alkyl products of dealkylation are conjugated with glutathione and further metabolized to mercapturic acid derivatives. Tin and its metabolites are excreted mainly in the urine and feces. (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | LD50: 44 mg/kg (Subcutaneous, Mouse) (8) LD50: 18 mg/kg (Intravenous, Rat) (8) LD50: 10 mg/kg (Intraperitoneal, Rabbit) (9) LD50: 10 mg/kg (Oral, Guinea pig) (9) LC50: 0.044 mg/L (Inhalation, Rat) (10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | 260 mg/kg for an adult human. (3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Fentin acetate is used as a fungicide. (7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Breathing or swallowing, or skin contact with organotins, can interfere with the way the brain and nervous system work, causing death in severe cases. Organic tin compounds may also damage the immune and reproductive system. (4, 5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Inorganic or organic tin compounds placed on the skin or in the eyes can produce skin and eye irritation. (5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||