Ozone (CHEM003683)
| Record Information | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2014-09-11 05:13:38 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-16 20:47:39 UTC | ||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM003683 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | |||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Ozone | ||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | ||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Ozone is a pale blue gas with a distinctively pungent smell. Ozone is formed from dioxygen by the action of ultraviolet light and also atmospheric electrical discharges. It is present in low concentrations throughout the Earth's atmosphere. In total, ozone makes up only 0.6 ppm of the atmosphere. In standard conditions, ozone is a pale blue gas that condenses at progressively cryogenic temperatures to a dark blue liquid and finally a violet-black solid. Ozone is a powerful oxidant (far more so than oxygen gas) and has many industrial and consumer applications related to oxidation. This same high oxidizing potential, however, causes ozone to damage mucus and respiratory tissues in animals, and also tissues in plants, above concentrations of about 100 ppb. Ozone's odor is sharp, reminiscent of chlorine, and detectable by many people at concentrations of as little as 10 ppb in air. Ozone, along with reactive forms of oxygen such as superoxide, singlet oxygen, hydrogen peroxide, and hypochlorite ions, is naturally produced by white blood cells. The largest use of ozone is in the preparation of pharmaceuticals, synthetic lubricants, and many other commercially useful organic compounds. It can also be used for bleaching substances and for killing microorganisms in air and water sources. Many municipal drinking water systems kill bacteria with ozone instead of the more common chlorine. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| ||||||||||||||||||||||||||||||||||||||||||||||||
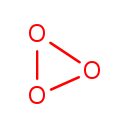
| Chemical Structure | |||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | O3 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 47.998 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 47.985 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 10028-15-6 | ||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | trioxirane | ||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | cyclic ozone | ||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | O1OO1 | ||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/O3/c1-2-3-1 | ||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | XQOAKYYZMDCSIA-UHFFFAOYSA-N | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | |||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of inorganic compounds known as other non-metal oxides. These are inorganic compounds containing an oxygen atom of an oxidation state of -2, in which the heaviest atom bonded to the oxygen belongs to the class of 'other non-metals'. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Inorganic compounds | ||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Homogeneous non-metal compounds | ||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Other non-metal organides | ||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Other non-metal oxides | ||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Other non-metal oxides | ||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | |||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | ||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Endogenous and Exogenous | ||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | |||||||||||||||||||||||||||||||||||||||||||||||||
| State | Gas | ||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Light blue gas. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | |||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | |||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Inhalation | ||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | Ozone is a strong oxidant and reacts directly with organic double bonds. When ozone breaks down to dioxygen it gives rise to oxygen free radicals, which are highly reactive and capable of damaging many organic molecules. Moreover, it is believed that the powerful oxidizing properties of ozone may be a contributing factor of inflammation. When inhaled, ozone reacts with compounds lining the lungs to form specific, cholesterol-derived metabolites that are thought to facilitate the build-up and pathogenesis of atherosclerotic plaques. These metabolites have been confirmed as naturally occurring in human atherosclerotic arteries and are categorized into a class of secosterols termed atheronals, generated by ozonolysis of cholesterol's double bond to form a 5,6 secosterol. Ozone has also been shown to form the suspected carcinogen bromate when it is introduced to source water with high bromide concentrations. Studies have also demonstrated an increase in collagen, a structural protein involved in fibrosis, following prolonged ozone exposure. Acute and short-term exposure studies have demonstrated ozone’s effects on lung lipids which include an increase in arachidonic acid (a pro-inflammatory molecule). | ||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Ozone can spontaneously break down to dioxygen. Most inhaled ozone reacts with cellular lipids and forms lipid peroxides which are eventually eliminated in the feces. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | 0.1 to 1 umol/mol will cause acute toxic effects. 40 nmol/mol will cause long term lung problems. Pulmonary edema developed in welders who had a severe acute exposure to an estimated 9 ppm ozone plus other air pollutants. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | 50 ppm for 60 minutes is expected to be fatal to humans. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity (not listed by IARC) (8). Tests in mice found no carcinogenic effect (7). | ||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Produced by white blood cells, generated by lightning, photocopiers and UV light. Used as an industrial chemical in the preparation of pharmaceuticals, synthetic lubricants. Used for bleaching and as a disinfectant. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Susceptible people can be adversely affected by ozone levels as low as 40 nmol/mol. In the EU, the current target value for ozone concentrations is 120 µg/m³ which is about 60 nmol/mol. In the US the recommended EPA standard is 75 nmol/mol. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | At acute levels, ozone is an irritant and lachrymator. At chronically low doses (~40 nmol/mol) there is a great deal of evidence to show that ground level ozone can harm lung function and irritate the respiratory system. Long-term exposure to ozone and the pollutants that produce it is linked to premature death, asthma, bronchitis, heart attacks and other cardiopulmonary problems. Long-term exposure to ozone has been shown to increase risk of death from respiratory illness. An 18-year study on 450,000 people in the US revealed that those living in cities with high ozone levels such as Houston or Los Angeles had an over 30% increased risk of dying from lung disease. Both acute and chronic exposures to ozone can negatively impact the barrier function of the lung, and these exposures can initiate an inflammatory response from the immune system. This immune response involves the release of biologically active mediators that can have adverse effects on the lung tissue itself. Some effects may include the thickening of the air-blood barrier in the lungs, thus reducing the diffusion of oxygen into the blood. Animals exposed to ozone produce and release high amounts of a sugar known as hyaluronan. Hyaluronan has been shown to be directly responsible for causing lung airways to narrow and become irritated. (6) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Exposure of 0.1 to 1 μmol/mol produces headaches, burning eyes and irritation to the respiratory passages. Exposure to 10 ppm ozone will cause pulmonary edema. Low doses act as a depressant and will induce sleep in certain individuals. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | EYES: Hold eye open and rinse slowly and gently with water for 15-20 minutes. Remove contact lenses, if present, after the first 5 minutes, then continue rinsing eye. Call a poison control center or doctor for treatment advice. INHALATION: Move person to fresh air. If person is not breathing, call 911 or an ambulance, then give artificial respiration, preferably by mouth-to-mouth if possible. Call a poison control center or doctor for further treatment advice. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | |||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | HMDB0035409 | ||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | FDB014085 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | C00034221 | ||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Ozone | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | 13375217 | ||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 25812 | ||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | 16206854 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||
| General References | |||||||||||||||||||||||||||||||||||||||||||||||||