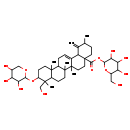
Identification Common Name 28-Glucosyl-3b,23-dihydroxy-12,19(29)-ursadien-28-oate 3-arabinoside Class Small Molecule Description 28-Glucosyl-3b,23-dihydroxy-12,19(29)-ursadien-28-oate 3-arabinoside is found in herbs and spices. 28-Glucosyl-3b,23-dihydroxy-12,19(29)-ursadien-28-oate 3-arabinoside is a constituent of Sanguisorba officinalis (burnet bloodwort). Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source 28-Glucosyl-3b,23-dihydroxy-12,19(29)-ursadien-28-Oic acid 3-arabinoside Generator 3,4,5-Trihydroxy-6-(hydroxymethyl)oxan-2-yl 9-(hydroxymethyl)-2,6a,6b,9,12a-pentamethyl-1-methylidene-10-[(3,4,5-trihydroxyoxan-2-yl)oxy]-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylic acid Generator
Chemical Formula C41 H64 O13 Average Molecular Mass 764.939 g/mol Monoisotopic Mass 764.435 g/mol CAS Registry Number 356785-73-4 IUPAC Name 3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl 9-(hydroxymethyl)-2,6a,6b,9,12a-pentamethyl-1-methylidene-10-[(3,4,5-trihydroxyoxan-2-yl)oxy]-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylate Traditional Name 3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl 9-(hydroxymethyl)-2,6a,6b,9,12a-pentamethyl-1-methylidene-10-[(3,4,5-trihydroxyoxan-2-yl)oxy]-2,3,4,5,6,7,8,8a,10,11,12,12b,13,14b-tetradecahydropicene-4a-carboxylate SMILES CC1CCC2(CCC3(C)C(=CCC4C5(C)CCC(OC6OCC(O)C(O)C6O)C(C)(CO)C5CCC34C)C2C1=C)C(=O)OC1OC(CO)C(O)C(O)C1O InChI Identifier InChI=1S/C41H64O13/c1-20-9-14-41(36(50)54-35-33(49)31(47)30(46)24(17-42)52-35)16-15-39(5)22(28(41)21(20)2)7-8-26-37(3)12-11-27(53-34-32(48)29(45)23(44)18-51-34)38(4,19-43)25(37)10-13-40(26,39)6/h7,20,23-35,42-49H,2,8-19H2,1,3-6H3 InChI Key LJCUOMZSVKPSBW-UHFFFAOYSA-N Chemical Taxonomy Description belongs to the class of organic compounds known as triterpene saponins. These are glycosylated derivatives of triterpene sapogenins. The sapogenin moiety backbone is usually based on the oleanane, ursane, taraxastane, bauerane, lanostane, lupeol, lupane, dammarane, cycloartane, friedelane, hopane, 9b,19-cyclo-lanostane, cycloartane, or cycloartanol skeleton. Kingdom Organic compounds Super Class Lipids and lipid-like molecules Class Prenol lipids Sub Class Terpene glycosides Direct Parent Triterpene saponins Alternative Parents Substituents Triterpene saponin
Triterpenoid
Steroid
Glycosyl compound
O-glycosyl compound
Monosaccharide
Oxane
Secondary alcohol
Carboxylic acid ester
Polyol
Acetal
Monocarboxylic acid or derivatives
Carboxylic acid derivative
Oxacycle
Organoheterocyclic compound
Hydrocarbon derivative
Primary alcohol
Alcohol
Organooxygen compound
Organic oxide
Organic oxygen compound
Carbonyl group
Aliphatic heteropolycyclic compound Molecular Framework Aliphatic heteropolycyclic compounds External Descriptors Not Available Spectra Spectra Spectrum Type Description Splash Key View Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_5) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_6) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_7) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TMS_1_8) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_4) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_5) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_6) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_7) - 70eV, Positive Not Available Spectrum Predicted GC-MS Predicted GC-MS Spectrum - GC-MS (TBDMS_1_8) - 70eV, Positive Not Available Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-0gbj-0400598600-cf0105f3bd3334ad8eaf Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-014i-0300947100-20cad6736a45fe161437 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-01b9-2200901000-ee9154f3961416d8826f Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-0il0-2300269800-1a9b7791c22bb963a273 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-0gx0-3700369400-b3ff8ae61bf1a93e882c Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-01ox-9500553000-440af4c4bdadf9ecce64 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Negative splash10-03di-0200003900-c31d2fd141aa6658a944 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Negative splash10-114u-3200229500-6b95a4bc8df6a23db33f Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Negative splash10-0a4l-9700552000-0524d62466b9a0d0d0d9 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 10V, Positive splash10-014i-0000469500-c0b7ffdea59885024f4b Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 20V, Positive splash10-0lds-1200932200-b899ac66b14042310aa6 Spectrum Predicted LC-MS/MS Predicted LC-MS/MS Spectrum - 40V, Positive splash10-0udi-2924500000-6d1fab46c87c0b737025 Spectrum