Dimenhydrinate (CHEM003519)
| Record Information | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Version | 1.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creation Date | 2014-08-30 21:04:18 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Update Date | 2026-04-13 20:40:07 UTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accession Number | CHEM003519 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Identification | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common Name | Dimenhydrinate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Small Molecule | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | Dimenhydrinate (Dramamine, Gravol and Vertirosan) is an over-the-counter drug used to prevent motion sickness. It is closely related to diphenhydramine HCl, or Benadryl. It is primarily a H1-antagonist, but also possesses an antimuscarinic effect. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Sources |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contaminant Type |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
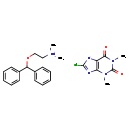
| Chemical Structure | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synonyms |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Formula | C24H28ClN5O3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Average Molecular Mass | 469.964 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monoisotopic Mass | 469.188 g/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Registry Number | 523-87-5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IUPAC Name | 8-chloro-1,3-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-7-ide; [2-(diphenylmethoxy)ethyl]dimethylazanium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Traditional Name | 8-chlorotheophylline(1-); [2-(diphenylmethoxy)ethyl]dimethylazanium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SMILES | CN1C2=C([N-]C(Cl)=N2)C(=O)N(C)C1=O.C[NH+](C)CCOC(C1=CC=CC=C1)C1=CC=CC=C1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Identifier | InChI=1S/C17H21NO.C7H7ClN4O2/c1-18(2)13-14-19-17(15-9-5-3-6-10-15)16-11-7-4-8-12-16;1-11-4-3(9-6(8)10-4)5(13)12(2)7(11)14/h3-12,17H,13-14H2,1-2H3;1-2H3,(H,9,10,13) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| InChI Key | DKHVTDUUNTVKOW-UHFFFAOYSA-N | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Taxonomy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description | belongs to the class of organic compounds known as diphenylmethanes. Diphenylmethanes are compounds containing a diphenylmethane moiety, which consists of a methane wherein two hydrogen atoms are replaced by two phenyl groups. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kingdom | Organic compounds | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Super Class | Benzenoids | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class | Benzene and substituted derivatives | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub Class | Diphenylmethanes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Direct Parent | Diphenylmethanes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alternative Parents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Substituents |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular Framework | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Descriptors |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Status | Detected and Not Quantified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Origin | Exogenous | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cellular Locations |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biofluid Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue Locations | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathways | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Applications | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biological Roles | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical Roles | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical Properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| State | Solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | White powder. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spectra |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Route of Exposure | Well absorbed after oral administration. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mechanism of Toxicity | The mechanism by which some antihistamines exert their antiemetic, anti–motion sickness, and antivertigo effects is not precisely known but may be related to their central anticholinergic actions. They diminish vestibular stimulation and depress labyrinthine function. An action on the medullary chemoreceptive trigger zone may also be involved in the antiemetic effect. Dimenhydrinate is a competitive antagonist at the histamine H1 receptor, which is widely distributed in the human brain. Dimenhydrinate's anti-emetic effect is probably due to H1 antagonism in the vestibular system in the brain. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | Hepatic (cytochrome P-450 system). Half Life: 1 to 4 hours | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Toxicity Values | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lethal Dose | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carcinogenicity (IARC Classification) | No indication of carcinogenicity to humans (not listed by IARC). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uses/Sources | Used for treating vertigo, motion sickness, and nausea associated with pregnancy. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimum Risk Level | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Health Effects | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms | Symptoms of overdose include delerium, hallucinations, and excitment. Patients may be violent and confused. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentrations | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Not Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| External Links | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DrugBank ID | DB00985 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HMDB ID | HMDB0015120 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FooDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phenol Explorer ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KNApSAcK ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BiGG ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BioCyc ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| METLIN ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikipedia Link | Dimenhydrinate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemspider ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ChEBI ID | 4604 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PubChem Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kegg Compound ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| YMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECMDB ID | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| References | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Synthesis Reference | Cusic, J.W.; U.S. Patent 2,499,058; February 28,1950; assigned to G.D. Searle & Co. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSDS | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General References | Not Available | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||